Fact Sheet
Technical brief: The importance of bioactivity in human milk and human milk–based products
Technical Brief: The Importance of Bioactivity in Human Milk and Human Milk–Based Products
Adequate, appropriate nutrition is vital for optimal growth and development in infants born prematurely. Human milk is a complex, dynamic, bioactive fluid with a myriad of compounds that make it the perfect nutrition for infants. Human milk also provides many protective factors against morbidity and mortality.1 Numerous studies have been conducted in an effort to identify the components of human milk and their influence on infant health and development. Many bioactive compounds are now recognized for their importance in immunity, growth, neurodevelopment, and overall long-term health as outlined in the table below.
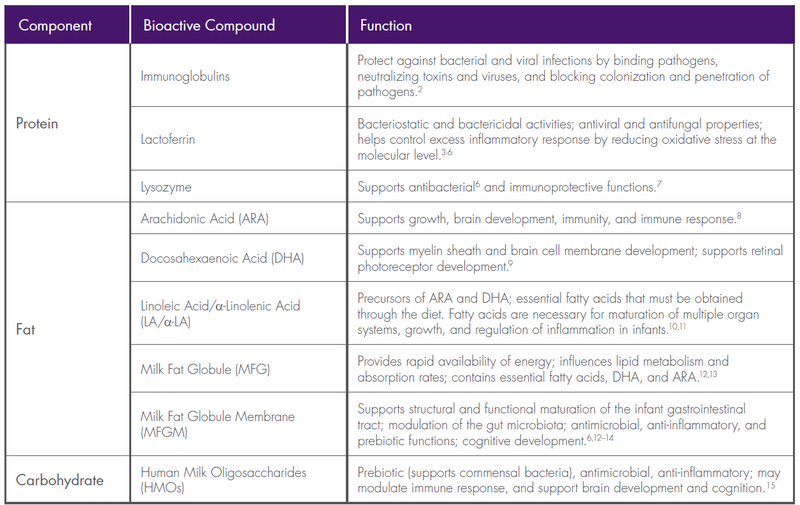
Table 2,3,4,5,6,7,8,9,10,11,12,13,14,15
Intake of human milk is especially critical for babies born prematurely, with mother’s own milk (MOM) being the best feeding option. In addition to supporting growth, development, and immunity, human milk has been shown to help mitigate the primary morbidities of prematurity including necrotizing enterocolitis (NEC), retinopathy of prematurity (ROP), bronchopulmonary dysplasia (BPD), and late-onset sepsis.16,17 There are often barriers to an adequate supply of MOM while premature infants are in the neonatal intensive care unit (NICU). Maternal/infant separation, the physical mechanics of pumping or hand expression, maternal illness, stress, and anxiety all play a role in the production of MOM.18
When an adequate amount of MOM is not available, the use of donor human milk is recommended.19 Similar to human blood and plasma, human milk carries with it the risk of pathogen transmission. To ensure donor human milk is safe for this vulnerable population, donor milk must undergo a pathogen inactivation process to remove potential transmissible microbiological contaminants before being given to infants in the NICU.
Pathogen Inactivation Methods
There are several methods of pathogen inactivation in use by donor milk providers. Each method has a different effect on the bioactivity of components found in human milk.
Vat and Holder Pasteurization: Heating milk to a set temperature to inactivate contaminants including bacteria and viruses. The process may reduce the activity and concentration of milk enzymes and some bioactive proteins.
Retort Sterilization: Commercial canning method that exposes milk to high temperature and pressure. This method inactivates bacteria and viruses and is shown to result in greater damage to bioactive proteins when compared to vat and Holder pasteurization.20
The Impact on Protein
Of the many nutrients in human milk, protein is of particular importance for growth, neurodevelopment, and immune response in premature infants. Human milk proteins are especially complex and susceptible to the various processing methods used in the dairy and food industries, including shearing (or pressure) and temperature. Since physiochemical properties of individual proteins differ, processing affects the proteins differently. For example, whey proteins, including immunoglobulins are heat labile and can be completely denatured with heat.21
The impact on proteins from the combination of pressure and temperature varies. When the shear rate (breaking down) and the temperature are both low, changes to the proteins are reversible. However, once the temperature exceeds 72°C and fragmentation and reformation of aggregates occurs, those changes result in permanent protein damage.22
A protein's function depends on its structure, therefore scrutiny must be given to human milk processing methods. Irreversible changes to protein structure, which likely occur with retort sterilization, will negatively impact biological function.
Damage to antibodies due to high temperature and pressure causes the loss of shape, removing the ability to recognize and bind to antigens, the protective feature antibodies typically provide.23 Studies suggest that some milk proteins are particularly resistant to proteolysis in the infant gastrointestinal tract, which may reflect the importance of these proteins remaining intact for non-nutritional bioactive function. By remaining intact, for example, secretory IgA (sIgA) can aid in development of the infant immune system and protect against infection.24
Human Milk Homogenization
Milk homogenization and heating milk to high temperatures, separately or in combination, cause structural and compositional changes to the MFGM and may play an important role in the decrease of MFGM bioactivity. Homogenized MFGMs lack structural integrity to exert functions in the digestive tract, where bioactivity is needed for gut health. Heating at 80°C (176°F) for 10 minutes has been shown to cause extensive protein denaturation and formation of supramolecular aggregates.25
Human milk gangliosides are important components of the MFGM with implicated bioactivities in neural and brain development, and maturation of the immune system and the gastrointestinal tract. While different heat treatments appear to have similar effects on the individual and total MFGM ganglioside content of human milk, homogenization in combination with heat treatment may have a negative impact. Specifically, Salcedo et al (2018) found that Holder pasteurization, high-temperature, short-time (HTST), ultrahigh temperature (UHT), and retort sterilization had a small but variable effect on the individual and total MFGM ganglioside content. However, the authors postulate that homogenization of milk may alter the digestibility and/or bioavailability of MFGM gangliosides due to the disruption of the MFGM structure. This release of gangliosides from the disrupted MFGM likely makes them more sensitive to heat treatment and may result in reduced biological activity.26
Effects of Processing on Bioactive Components
In 2018, the Journal of Human Lactation published study results comparing the macronutrient (protein, carbohydrate, fat), immune-protective protein, and HMO content of human milk from three independent milk banks that use pasteurization (Holder vs vat techniques) or retort sterilization. Results showed the concentrations of protein and fat were significantly (P < .05) less in the retort sterilized samples compared with the Holder and vat pasteurized samples, respectively. The concentrations of IgA, Immunoglobulin G, Immunoglobulin M, lactoferrin, lysozyme, α-lactalbumin, casein, osteopontin, and α-1-antitrypsin proteins were significantly (P < .05) less in the retort sterilized samples compared with vat and/or Holder pasteurized samples. The total HMO concentrations with vat pasteurization were similar to the human milk reference and also the concentration of the HMOs containing fucose, sialic acid, and nonfucosylated neutral sugars were significantly (P < .05) less in retort sterilized compared with vat and Holder pasteurized samples.20
What is the take-away?
Bioactivity matters and MOM is best. But when enough MOM is not available, it is vital to select the right human donor milk and/or human milk–based product. To ensure the preterm infants under your care have the best possible outcomes, select a product that has been extensively evaluated in multiple clinical studies with results published in peer-reviewed journals. Understanding the effects of the different processing methods on bioactivity will help you make a clinically sound decision when nurturing your fragile patients.
References
1 Gila-Diaz A, Arribas SM, Algara A, Martín-Cabrejas MA, López de Pablo ÁL, Sáenz de Pipaón M, Ramiro-Cortijo D. A review of bioactive factors in human breastmilk: a focus on prematurity. Nutrients. 2019;11(6):1307. doi:10.3390/nu11061307
2 Breedveld A, van Egmond M. IgA and FcαRI: pathological roles and therapeutic opportunities. Front Immunol. 2019;10:553. doi:10.3389/fimmu.2019.00553
3 Manzoni P. Clinical studies of lactoferrin in neonates and infants: an update. Breastfeed Med. 2019;14(Suppl 1):S25-S27. doi:10.1089/bfm.2019.0033
4 Kell DB, Heyden EL, Pretorius E. The biology of lactoferrin, an iron-binding protein that can help defend against viruses and bacteria. Front Immunol. 2020;11:1221. doi:10.3389/fimmu.2020.01221
5 Actor JK, Hwang SA, Kruzel ML. Lactoferrin as a natural immune modulator. Curr Pharm Des. 2009;15(17):1956-1973. doi:10.2174/138161209788453202
6 Lönnerdal B. Bioactive proteins in human milk—potential benefits for preterm infants. Clin Perinatol. 2017;44(1):179-191. doi:10.1016/j.clp.2016.11.013
7 Peila C, Moro GE, Bertino E, et al. The effect of holder pasteurization on nutrients and biologically-active components in donor human milk: a review. Nutrients. 2016;8(8):477. doi:10.3390/nu8080477
8 Hadley KB, Ryan AS, Forsyth S, Gautier S, Salem N Jr. The essentiality of arachidonic acid in infant development. Nutrients. 2016;8(4):216. doi:10.3390/nu8040216
9 Bzikowska-Jura A, Czerwonogrodzka-Senczyna A, Jasińska-Melon E, et al. The concentration of omega-3 fatty acids in human milk is related to their habitual but not current intake. Nutrients. 2019;11(7):1585. doi:10.3390/nu11071585
10 Gleason CA, Juul SE, eds. Avery's diseases of the newborn. 10th ed. Elsevier:Philadelphia, PA;2018:1009-1022.e4.
11 Fleig LN, Unger JP, Cluette-Brown JE, et al. Longitudinal fatty acid levels in preterm infants fed
12 Lee H, Padhi E, Hasegawa Y, et al. Compositional dynamics of the milk fat globule and its role in infant development. Front Pediatr. 2018;6:313. doi:10.3389/fped.2018.00313
13 Weiser MJ, Butt CM, Mohajeri MH. Docosahexaenoic acid and cognition throughout the lifespan. Nutrients. 2016;8(2):99. doi:10.3390/nu8020099
14 Gallier S, Rowan A. Breastmilk composition and brain development. White paper. Accessed January 8, 2021. https://myaccount.nzmp.com/sfc/servlet.shepherd/version/download/ 0682s000001RSPOAA4
15 Bode L. Human milk oligosaccharides: every baby needs a sugar mama. Glycobiology. 2012;22(9):1147-1162. doi:10.1093/glycob/cws074
16 Hair AB, Peluso AM, Hawthorne KM, et al. Beyond necrotizing enterocolitis prevention: improving outcomes with an exclusive human milk-based diet [published correction appears in Breastfeed Med. 2017;12(10):663] Breastfeed Med. 2016;11(2):70-74. doi:10.1089/bfm.2015.0134
17 Delaney Manthe E, Perks PH, Swanson JR. Team-based implementation of an exclusive human milk diet. Adv Neonatal Care. 2019;19(6):460-467. doi:10.1097/ANC.0000000000000676
18 Fernández Medina IM, Fernández-Sola C, López-Rodríguez MM, Hernández-Padilla JM, Jiménez Lasserrotte MDM, Granero-Molina J. Barriers to providing mother’s own milk to extremely preterm infants in the NICU. Adv Neonatal Care. 2019;19(5):349-360. doi:10.1097/ANC.0000000000000652
19 Kleinman RE, Greer FR, eds. American Academy of Pediatrics Committee on Nutrition. Pediatric Nutrition. 8th ed. Itasca, IL: American Academy of Pediatrics; 2019.
20 Meredith-Dennis L, Xu G, Goonatilleke E, Lebrilla CB, Underwood MA, Smilowitz JT. Composition and variation of macronutrients, immune proteins, and human milk oligosaccharides in human milk from nonprofit and commercial milk banks. J Hum Lact. 2018;34(1):120-129. doi:10.1177/0890334417710635
21 Patel H, Patel S. Technical Report: Understanding the Role of Dairy Proteins in Ingredient and Product Performance. Dairy Protein Report, 2015.
22 Mediwaththe A, Bogahawaththa D, Grewal MK, Chandrapala J, Vasiljevic T. Structural changes of native milk proteins subjected to controlled shearing and heating. Food Res Int. 2018;114:151-158. doi:10.1016/j.foodres.2018.08.001
23 Goyal A, Sharma V, Upadhyay N, Sihag M, Kaushik R. High pressure processing and its impact on milk proteins: a review. Res Rev: J Dairy Sci Technol. 2013;2(1):12-20. Accessed March 15, 2021. http://sciencejournals.stmjournals.in/index.php/RRJoDST/article/view/848
24 Dallas DC, Underwood MA, Zivkovic AM, German JB. Digestion of protein in premature and term infants. J Nutr Disord Ther. 2012;2(3):112. doi:10.4172/2161-0509.1000112
25 Zanabria R, Tellez AM, Griffiths MW, et al. The antiproliferative properties of the milk fat globule membrane are affected by extensive heating. Dairy Sci Technol. 2014;94:439–453. doi:10.1007/s13594-014-0171-8
26 Salcedo J, Karav S, Le Parc A, et al. Application of industrial treatments to donor human milk: influence of pasteurization treatments, storage temperature, and time on human milk gangliosides. npj Sci Food. 2018;2:5. doi:10.1038/s41538-018-0013-9
